Meta Description
Learn how lead acid batteries work, including chemical reactions, charging and discharging process, and key components.
Keywords
lead acid battery working principle, how lead acid battery works, battery chemical reaction, energy storage battery
Introduction
Lead acid batteries are one of the oldest and most widely used energy storage technologies. Despite the emergence of newer battery types, they remain essential in solar energy storage, telecom backup, and industrial applications.
Understanding how lead acid batteries work helps users:
- Improve system design
- Extend battery lifespan
- Optimize charging and usage
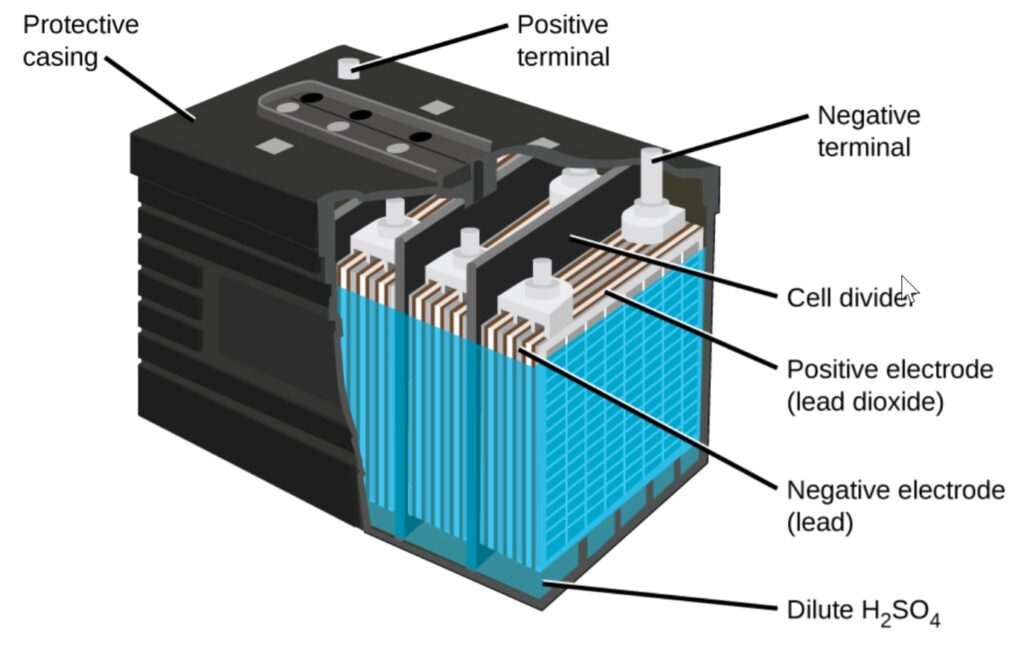
Basic Components of a Lead Acid Battery
A typical lead acid battery consists of:
- Positive plate (lead dioxide, PbO₂)
- Negative plate (lead, Pb)
- Electrolyte (sulfuric acid, H₂SO₄)
- Separator
- Battery container
How Lead Acid Batteries Generate Electricity
The battery works through electrochemical reactions.
Discharge Process
When the battery is in use:
- Lead dioxide reacts with sulfuric acid
- Lead reacts with sulfuric acid
- Both plates form lead sulfate (PbSO₄)
At the same time:
👉 Electrical energy is released
Charging Process
When charging:
- Lead sulfate converts back to lead dioxide and lead
- Sulfuric acid concentration increases
👉 Energy is stored again
Chemical Reaction Summary
Discharge reaction:
PbO₂ + Pb + 2H₂SO₄ → 2PbSO₄ + 2H₂O
Charging is the reverse reaction.
Why Sulfation Occurs
Sulfation happens when:
- Battery remains in a discharged state
- Lead sulfate crystals harden
👉 This reduces battery capacity.
Factors Affecting Performance
- Temperature
- Depth of discharge
- Charging method
- Maintenance
Applications
Lead acid batteries are widely used in:
- Solar energy storage
- UPS systems
- Telecom backup
- Industrial power systems
Conclusion
Lead acid batteries operate through reversible chemical reactions that allow them to store and release energy efficiently.
👉 Understanding these principles helps users maximize performance and lifespan.
FAQ
Q1: Why does battery lose capacity?
Due to sulfation and aging.
Q2: Can sulfation be reversed?
Partially, with proper charging.